Soap Chemical Bond . Its fundamental chemistry involves the combination. how soap works is due to its unique chemistry, the hydrophilic (loves water) and hydrophobic (hates water) parts of soap act to combine soapy water with grease, dirt, or oil. C=c bonds cause the fatty. soap molecules have a hybrid structure, with a hydrophilic head that bonds to water and a hydrophobic tail that avoids. This combination creates clusters of soap, water, and grime called micelles. Unsaturated fatty acids contain double and/or triple bonds. soaps act as cleansers because the two ends of a soap molecule are so different. Soap is a classic cleaning agent that has been used for centuries. if the ph of a soap solution is lowered by acidic contaminants, insoluble fatty acids precipitate and form a scum.
from stock.adobe.com
Soap is a classic cleaning agent that has been used for centuries. how soap works is due to its unique chemistry, the hydrophilic (loves water) and hydrophobic (hates water) parts of soap act to combine soapy water with grease, dirt, or oil. soaps act as cleansers because the two ends of a soap molecule are so different. if the ph of a soap solution is lowered by acidic contaminants, insoluble fatty acids precipitate and form a scum. Its fundamental chemistry involves the combination. soap molecules have a hybrid structure, with a hydrophilic head that bonds to water and a hydrophobic tail that avoids. This combination creates clusters of soap, water, and grime called micelles. Unsaturated fatty acids contain double and/or triple bonds. C=c bonds cause the fatty.
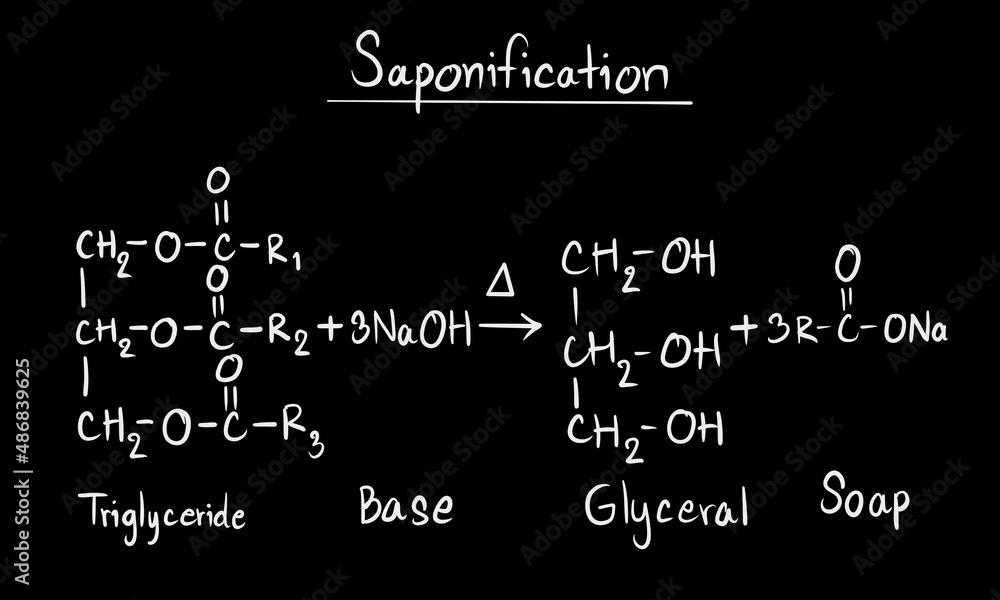
Saponification equation, reaction of soap, chemistry equation of soap
Soap Chemical Bond Soap is a classic cleaning agent that has been used for centuries. This combination creates clusters of soap, water, and grime called micelles. soaps act as cleansers because the two ends of a soap molecule are so different. if the ph of a soap solution is lowered by acidic contaminants, insoluble fatty acids precipitate and form a scum. Its fundamental chemistry involves the combination. Soap is a classic cleaning agent that has been used for centuries. C=c bonds cause the fatty. Unsaturated fatty acids contain double and/or triple bonds. how soap works is due to its unique chemistry, the hydrophilic (loves water) and hydrophobic (hates water) parts of soap act to combine soapy water with grease, dirt, or oil. soap molecules have a hybrid structure, with a hydrophilic head that bonds to water and a hydrophobic tail that avoids.
From ffden-2.phys.uaf.edu
Soap and Water Interaction Soap Chemical Bond Soap is a classic cleaning agent that has been used for centuries. soap molecules have a hybrid structure, with a hydrophilic head that bonds to water and a hydrophobic tail that avoids. Unsaturated fatty acids contain double and/or triple bonds. C=c bonds cause the fatty. soaps act as cleansers because the two ends of a soap molecule are. Soap Chemical Bond.
From www.thoughtco.com
How Saponification Makes Soap Soap Chemical Bond if the ph of a soap solution is lowered by acidic contaminants, insoluble fatty acids precipitate and form a scum. This combination creates clusters of soap, water, and grime called micelles. Soap is a classic cleaning agent that has been used for centuries. Unsaturated fatty acids contain double and/or triple bonds. how soap works is due to its. Soap Chemical Bond.
From www.pinterest.com
Cleansing Action Of Soap. Soap, Cleanse, Molecules Soap Chemical Bond C=c bonds cause the fatty. Unsaturated fatty acids contain double and/or triple bonds. how soap works is due to its unique chemistry, the hydrophilic (loves water) and hydrophobic (hates water) parts of soap act to combine soapy water with grease, dirt, or oil. Soap is a classic cleaning agent that has been used for centuries. This combination creates clusters. Soap Chemical Bond.
From www.thoughtco.com
How Soap Works Soap Chemical Bond Its fundamental chemistry involves the combination. Unsaturated fatty acids contain double and/or triple bonds. Soap is a classic cleaning agent that has been used for centuries. This combination creates clusters of soap, water, and grime called micelles. soap molecules have a hybrid structure, with a hydrophilic head that bonds to water and a hydrophobic tail that avoids. soaps. Soap Chemical Bond.
From cbse.myindialist.com
Chemistry X Carbon and its Compounds SOAPS AND DETERGENTS CBSE Soap Chemical Bond how soap works is due to its unique chemistry, the hydrophilic (loves water) and hydrophobic (hates water) parts of soap act to combine soapy water with grease, dirt, or oil. This combination creates clusters of soap, water, and grime called micelles. soaps act as cleansers because the two ends of a soap molecule are so different. soap. Soap Chemical Bond.
From www.dreamstime.com
General Formula of Solid and Liquid Soap Molecule. RCOONa, RCOOK Soap Chemical Bond soaps act as cleansers because the two ends of a soap molecule are so different. This combination creates clusters of soap, water, and grime called micelles. Its fundamental chemistry involves the combination. if the ph of a soap solution is lowered by acidic contaminants, insoluble fatty acids precipitate and form a scum. C=c bonds cause the fatty. . Soap Chemical Bond.
From ar.inspiredpencil.com
Soap Molecule Polar Or Nonpolar Soap Chemical Bond Unsaturated fatty acids contain double and/or triple bonds. soaps act as cleansers because the two ends of a soap molecule are so different. C=c bonds cause the fatty. soap molecules have a hybrid structure, with a hydrophilic head that bonds to water and a hydrophobic tail that avoids. how soap works is due to its unique chemistry,. Soap Chemical Bond.
From www.alamy.com
General formula of solid and liquid soap molecule. RCOONa, RCOOK Soap Chemical Bond if the ph of a soap solution is lowered by acidic contaminants, insoluble fatty acids precipitate and form a scum. Its fundamental chemistry involves the combination. C=c bonds cause the fatty. Unsaturated fatty acids contain double and/or triple bonds. how soap works is due to its unique chemistry, the hydrophilic (loves water) and hydrophobic (hates water) parts of. Soap Chemical Bond.
From www.sharpestarena.com
Production of Soap Complete Project on Soap Making Soap Chemical Bond if the ph of a soap solution is lowered by acidic contaminants, insoluble fatty acids precipitate and form a scum. This combination creates clusters of soap, water, and grime called micelles. Its fundamental chemistry involves the combination. soap molecules have a hybrid structure, with a hydrophilic head that bonds to water and a hydrophobic tail that avoids. Unsaturated. Soap Chemical Bond.
From www.youtube.com
Chemistry 101 How does soap work? YouTube Soap Chemical Bond C=c bonds cause the fatty. This combination creates clusters of soap, water, and grime called micelles. if the ph of a soap solution is lowered by acidic contaminants, insoluble fatty acids precipitate and form a scum. soap molecules have a hybrid structure, with a hydrophilic head that bonds to water and a hydrophobic tail that avoids. soaps. Soap Chemical Bond.
From app.emaze.com
Presentation Name on emaze Soap Chemical Bond how soap works is due to its unique chemistry, the hydrophilic (loves water) and hydrophobic (hates water) parts of soap act to combine soapy water with grease, dirt, or oil. Its fundamental chemistry involves the combination. Soap is a classic cleaning agent that has been used for centuries. soaps act as cleansers because the two ends of a. Soap Chemical Bond.
From www.youtube.com
How Soap Works and Chemical Bonds YouTube Soap Chemical Bond This combination creates clusters of soap, water, and grime called micelles. C=c bonds cause the fatty. Unsaturated fatty acids contain double and/or triple bonds. how soap works is due to its unique chemistry, the hydrophilic (loves water) and hydrophobic (hates water) parts of soap act to combine soapy water with grease, dirt, or oil. if the ph of. Soap Chemical Bond.
From www.slideserve.com
PPT Making Soap PowerPoint Presentation, free download ID1971971 Soap Chemical Bond soaps act as cleansers because the two ends of a soap molecule are so different. Unsaturated fatty acids contain double and/or triple bonds. how soap works is due to its unique chemistry, the hydrophilic (loves water) and hydrophobic (hates water) parts of soap act to combine soapy water with grease, dirt, or oil. soap molecules have a. Soap Chemical Bond.
From exoxmnoxz.blob.core.windows.net
How Does Soap Work Science at Kelly Kline blog Soap Chemical Bond Soap is a classic cleaning agent that has been used for centuries. Unsaturated fatty acids contain double and/or triple bonds. soap molecules have a hybrid structure, with a hydrophilic head that bonds to water and a hydrophobic tail that avoids. soaps act as cleansers because the two ends of a soap molecule are so different. This combination creates. Soap Chemical Bond.
From bossanovasoap.com
CHEMISTRY Soap Chemical Bond Its fundamental chemistry involves the combination. Soap is a classic cleaning agent that has been used for centuries. if the ph of a soap solution is lowered by acidic contaminants, insoluble fatty acids precipitate and form a scum. soaps act as cleansers because the two ends of a soap molecule are so different. soap molecules have a. Soap Chemical Bond.
From www.slideserve.com
PPT Soap Describe how soap is made from fatty acids and alkalis Soap Chemical Bond how soap works is due to its unique chemistry, the hydrophilic (loves water) and hydrophobic (hates water) parts of soap act to combine soapy water with grease, dirt, or oil. C=c bonds cause the fatty. Soap is a classic cleaning agent that has been used for centuries. soaps act as cleansers because the two ends of a soap. Soap Chemical Bond.
From cosmosmagazine.com
The chemistry of soap Soap Chemical Bond how soap works is due to its unique chemistry, the hydrophilic (loves water) and hydrophobic (hates water) parts of soap act to combine soapy water with grease, dirt, or oil. Unsaturated fatty acids contain double and/or triple bonds. soap molecules have a hybrid structure, with a hydrophilic head that bonds to water and a hydrophobic tail that avoids.. Soap Chemical Bond.
From www.slideserve.com
PPT How Does Soap Work? PowerPoint Presentation, free download ID Soap Chemical Bond soaps act as cleansers because the two ends of a soap molecule are so different. This combination creates clusters of soap, water, and grime called micelles. Unsaturated fatty acids contain double and/or triple bonds. C=c bonds cause the fatty. soap molecules have a hybrid structure, with a hydrophilic head that bonds to water and a hydrophobic tail that. Soap Chemical Bond.